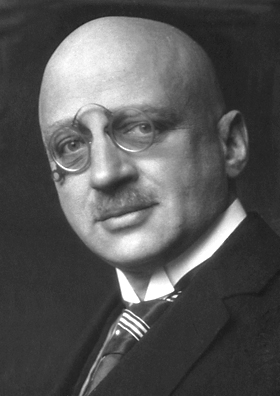
Fritz Haber was on hand to witness the effects of his chlorine gas weapon on Algerian and Canadian troops at the Second Battle of Ypres in April 1915. [Nobel Foundation/Wikimedia]
The cloud was, in fact, more than 160 tonnes of poisonous chlorine gas and as it rolled over the French colonials on the Canadians’ left flank, the Algerian soldiers began choking and gasping for air. Some turned and ran, but the gas followed them. The nearest Algerians made for the Canadian trenches across the road.
“Our trenches were shortly filled with them crowding in from out left,” said Canadian Lieutenant-Colonel Ian Sinclair of the 13th (Royal Highlanders of Canada) Battalion. “They were mostly blind and choking to death, and as fast as they died were just heaved behind the trench.”
The attack during the Second Battle of Ypres—the first successful incidence of modern chemical warfare—had the desired effect, opening a 6.5-kilometre gap in the Allied line, which the Canadians promptly filled as best they could. Rather than the breakthrough the Germans desired, the enemy’s advance stopped short, handicapped by a lack of adequate reserves due to fighting on the Eastern Front and the ill-equipped troops’ own distrust of their new weapon.

A surviving WW I gas shell sits in a classroom display at the Belgian military bomb disposal unit, DOVO.The chlorine gas used at Ypres was deployed through tubes from cylinders in the trenches. Artillery shells like this were used later in the war.’
[Stephen J. Thorne/Legion Magazine]
“The Canadians…got badly gassed,” recalled Bert Newman of the Royal Army Medical Corps. “In the end you could see all these poor chaps laying on the Menin Road, gasping for breath. And the thing was it was no gas masks then, you see, and a lot of these chaps just had to wet their handkerchiefs and put it over their mouth or do what they could.”
A British sergeant-major gathered several big jars of Vaseline and applied it to the victims’ throats, but it was of little help.
“On the front field one can see the dead lying here and there, and in places where an assault has been they lie very thick on the front slopes of the German trenches,” Canadian Lieutenant-Colonel John McRae wrote less than two weeks before he authored the poem “In Flanders Fields” after the death of a close comrade.
In their first two weeks of fighting around Ypres, the Canadians suffered some 6,000 casualties, a third of the division. A thousand lay dead on the battlefield, many of them gas victims.

The St. Julien Canadian Memorial, commonly known as the Brooding Soldier, memorializes the Second Battle of Ypres and overlooks the site of the first gas attack. [Stephen J. Thorne/Legion Magazine]
Fritz Haber, the German scientist who came up with the diabolical weapon, is a study in contradiction, a Jew who converted to Christianity in 1892 to further his career, whose work not only caused misery and death to countless numbers of people in two world wars, but also saved—and continues to save—countless millions. A scientific institute in Israel bears his name.
In 1918, three years after his weapon was introduced on the battlefield at Ypres, Haber was awarded the Nobel Prize in Chemistry for developing the Haber-Bosch process, a method of synthesizing ammonia from nitrogen and hydrogen that remains fundamental to the large-scale synthesis of fertilizers—and explosives.
(Upon his death in 1896, Swedish chemist and engineer Alfred Nobel, whose most famous invention, dynamite, was already responsible for millions of deaths, created and donated his entire fortune to the Nobel Prize institution, which would annually recognize those who “conferred the greatest benefit to humankind.”)

Haber’s first wife, Clara Immerwahr, was a trailblazing scientist and women’s rights activist. She died by suicide 10 days after the first chlorine gas attack at Ypres. [Wikimedia]
The ability to produce far larger quantities of nitrogen-based fertilizers in turn supported much greater agricultural yields. Today, the annual world production of synthetic nitrogen fertilizer based on the Haber-Bosch process exceeds 100 million tonnes and the food base of half the world’s population depends on it.
Yet Haber was a fanatical German nationalist whose work in developing chemical weapons is said to have contributed to the May 1915 suicide of his first wife, trailblazing scientist and women’s rights activist Clara Immerwahr.
He continued his work on chemical weapons even after the 1918 armistice. Two of his children killed themselves following the Second World War, before which Zyklon A, a pesticide developed at Haber’s institute, spawned Zyklon B, the chemical the Nazis used to exterminate more than a million Jews and other “undesirables” in the 1940s.
In a not-so-improbable twist of fate, several members of Haber’s extended family died in Nazi concentration camps, including his half-sister Frieda’s daughter, Hilde Glücksmann, her husband and their two children.
Despite his contributions to the German cause, his Jewish ancestry caught up with him and, after 1933, Haber lived out is life in exile.

British artist William Roberts depicted the German gas attack on French colonial troops outside Ypres on April 22, 1915.
[William Roberts/National Gallery of Canada/8729]
Other gases had failed to do the job, so it was left to Haber to come up with a solution. He immediately assembled a team of more than 150 scientists and 1,300 technical personnel.
A special gas-warfare troop was formed (Pioneer Regiments 35 and 36) under the command of Otto Peterson. Haber and Friedrich Kerschbaum served as advisers. Haber recruited physicists, chemists and other scientists.
Future Nobel laureates James Franck, Gustav Hertz (both physics) and Otto Hahn (chemistry) served as gas troops in Haber’s unit. Haber seized on chlorine gas largely because it was heavy and settled downward. His teams also developed other deadly gases for use in trench warfare.

A reconstructed Allied trench at the Passchendaele Museum, housed on the onetime property of a Belgian family in Zonnebeke.
[Stephen J. Thorne/Legion Magazine]
“The disapproval that the knight had for the man with the firearm is repeated in the soldier who shoots with steel bullets towards the man who confronts him with chemical weapons,” he said. “The gas weapons are not at all more cruel than the flying iron pieces; on the contrary, the fraction of fatal gas diseases is comparatively smaller, the mutilations are missing.”
In his studies of the weapon’s effects, Haber noted that exposure to a low concentration for a long time often had the same effect—death—as exposure to a high concentration for a short time. He formulated a simple mathematical relationship between the gas concentration and the necessary exposure time. This relationship became known as Haber’s rule.
Haber died in January 1934, at just 65 years old. He remained the object of much criticism for his involvement in developing chemical weapons, both from contemporaries, especially Albert Einstein, and modern-day scientists.
Yet in 1981, the Minerva Foundation of the Max Planck Society and the Hebrew University of Jerusalem established the Fritz Haber Research Center for Molecular Dynamics, based at the Institute of Chemistry of the Hebrew University. Its purpose is the promotion of Israeli-German scientific collaboration in the field of molecular dynamics. The centre also houses the Fritz Haber Library.
—
This is the second in a series of columns on Flanders. Next week: The standoff at Ypres and the rise of stagnant warfare.
Advertisement












